EU MDR compliance support for a Global Medical Device Manufacturer
UDI Consulting Support for a Global Medical Device Company
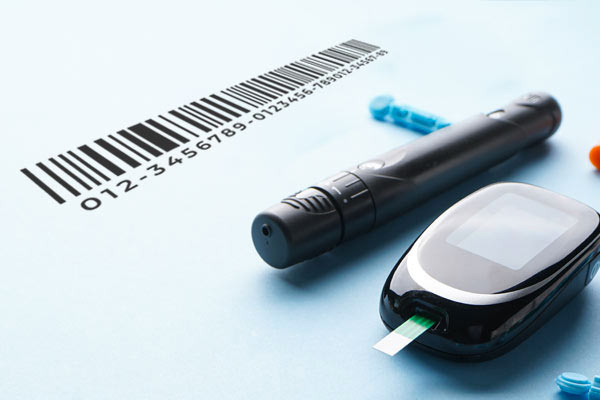
Project Summary
A leading global medical device company needed assistance from Celegence to support their Unique Device Identification (UDI) compliance labeling requirements. The client required assistance establishing both the UDIs and GTINs, as well as updating the GUDID database in compliance for the US and EU regions for their entire product portfolio.
The manufacturer required a unique product label in barcode format on every device. This UDI label included pertinent information such as the manufactured date and lot number. Maintaining efficient data management was also a prominent obstacle for the client.
Celegence Solution & Approach:
Celegence conducted a gap analysis through a workshop to outline the necessary steps to prepare the Unique Device Identifier (UDI) for the US and EU regions. The key activities included:
Highlights:
A remote workshop to assess the customer’s current regulatory labeling process
Determination of the packaging levels
Defining the product details in GS1 for the UDI-DI and generate the BUDI-DI
Update the necessary details in the GUDID database
Advisory consulting related to their portfolio of 21 products
Project Achievements
Celegence continues to ensure the customer’s processes comply with the UDI requirements. Some of the key deliverables included:
A report detailing the current UDI status in GS1 and the GUDID Database
Project called for UDI reporting in the EUDAMED database once available, expected to commence in June of 2022
100% achievement in quality & delivery performance despite tight timelines, an achievement due to Celegence’s proven procedures and practices
Project Success
The key to the continued success of this partnership is the involvement of specialized resources with the appropriate therapeutic area expertise working in tandem with the client’s regulatory team, and together, the two teams delivered multiple documents in expedited timelines. Celegence, as a leading regulatory affairs service provider can act as your Authorized Representative (AR), register your medical devices in EUDAMED, and can assist your organization with registering your own UDIs for your medical devices..
If your Regulatory Affairs
team needs assistance on maintaining UDI compliance of a current device, or are looking to implement a UDI system for a new device, our expert team is here to help. Get in touch today to discuss your regulatory needs by reaching out to info@celegence.com. Find out more information on Celegence’s Medical Device capabilities.
Learn MoreOther Related Articles

27 Jun, 2025
Contact Us Today
From document publishing automation to eCTD submissions and beyond, Celegence is your trusted partner for regulatory affairs excellence. Contact us to learn how we can help you achieve your compliance goals efficiently and cost-effectively.
"*" indicates required fields