"*" indicates required fields
EMA Roadmap Update: PMS Capabilities and the End of the Legacy Era

01 Apr, 2026
Is your Portfolio “FAIR” enough for a FHIR submission
Are you ready? Assess your gaps with our PMS Readiness Scorecard:
- SPOR Alignment
- Data Extraction Completion
- Technical Format/FHIR R5 Export
- MBO/Maintenance Process Model
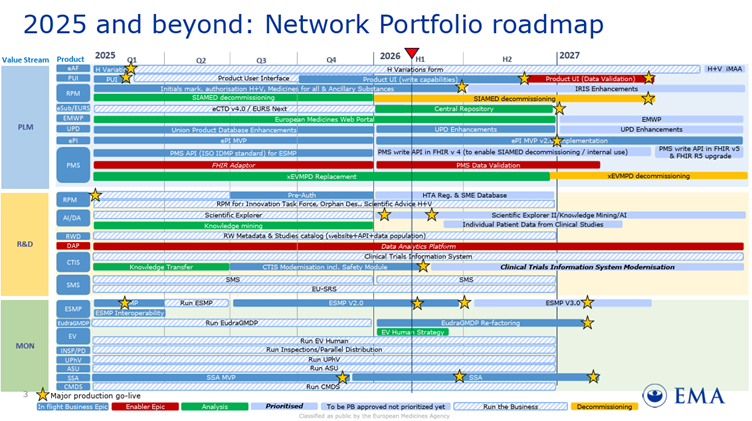
For years, we’ve talked about the “future” of ISO IDMP. As of the latest EMA Network Portfolio Roadmap (Updated March 2026), that future has a firm timeline.
The era of siloed, narrative-driven records is closing. The EMA is officially moving toward a unified “Digital Twin” model of our medicinal portfolios, and two legacy giants are now on the decommissioning block.
The Decommissioning Roadmap
According to the EMA’s strategic roadmap, the transition away from legacy systems is already in flight:
- SIAMED Decommissioning : The phase-out of SIAMED (the legacy system for managing centrally authorised products) is a critical milestone. The roadmap indicates that the transition is timed to follow the migration of SIAMED data into the PMS (Product Management Service).
- xEVMPD Decommissioning : The decommissioning of the eXtended EudraVigilance Medicinal Product Dictionary (xEVMPD) is explicitly scheduled to take place as the PMS API FHIR v5.0 reaches full write capabilities.
From “Records” to “Data Objects”
This isn’t just a technical swap; it’s a fundamental pivot in the Regulatory Affairs Operating Model:
- SIAMED → IRIS & PMS: Regulatory procedure management is shifting to IRIS, while product data moves into the PMS.
- xEVMPD → ISO-IDMP (PMS): The static Article 57 entries we’ve managed for decades are being replaced by high-fidelity, structured data objects based on HL7 FHIR standards.
The Strategic Link : Why FAIR is the Foundation for FHIR
While FHIR (Fast Healthcare Interoperability Resources) is the format for exchange, FAIR is the standard for the data itself. Without FAIR principles, the move to FHIR is merely “digitizing a mess.”
- Findable: Beyond “File Names” to Persistent Identifiers
In the legacy world, a product was found by searching for a PDF in a folder. In the IDMP era, “Findable” means every substance, organization, and product has a Globally Unique and Persistent Identifier (PID).
- Operational Challenge : Mapping legacy data to OMS Loc-IDs and SMS UUIDs.
- Remediation Goal : Ensure every data element in your RIM system is tagged with EMA-compliant referential IDs so the PLM Portal can “find” and pre-populate your forms.
- Accessible: Standardized Communication Protocols
Accessibility in FAIR doesn’t mean “open to everyone,” but rather that data is retrievable via standardized protocols (like HTTPS) with clear authentication.
- The FHIR Connection : The PMS API uses FHIR over HTTPS, allowing for secure, machine-to-machine “write” endpoints.
- Remediation Goal : Transition from manual portal entry to automated API-based submissions that ensure your data is always accessible for real-time validation.
- Interoperable : Speaking the Same Language
This is the heart of the IDMP transition. Interoperability requires the use of Controlled Vocabularies (referentials) and standardized data models.
- The FHIR Connection : FHIR resources provide the common structure (the “grammar”), while IDMP-compliant ontologies (like those developed by the Pistoia Alliance) provide the “vocabulary”.
- Remediation Goal : Cleanse “Dark Data” (narrative text in SmPCs) and convert it into structured, machine-readable formats that can be integrated across PV, Quality, and Supply Chain systems.
- Reusable : The Ultimate ROI of IDMP
Data is reusable only if it has rich metadata and clear provenance (history of where the data came from).
- Operational Challenge : Establishing a “Single Source of Truth” where a change in a manufacturing site automatically updates every related product record.
- Remediation Goal : Move away from “one-and-done” submission prep. By making data FAIR, you can reuse it for ePI (electronic Product Information), shortages monitoring, and automated safety reporting.
The 2026 Roadmap: From Remediation to Submission
| Phase | Milestone | Focus Area |
| Phase 1: Remediation | Ongoing | Extracting data from SmPCs; Mapping to SPOR referential using FAIR principles. |
| Phase 2: Validation | Current | Testing data against the January 2026 portal business rules to avoid technical rejections. |
| Phase 3: Submission | June 2026 | Mandatory FHIR-structured enrichment for ULCM products via the PMS API. |
| Phase 4: Optimization | Dec 2026+ | Extending FAIR data across the full portfolio and internal cross-functional systems. |
Are You Still Filing Records, or Are You Managing Data?
To help you identify where your team stands, Download your Free IDMP & PMS Compliance Checklist.
Conclusion: Compliance as a Competitive Edge
IDMP remediation is a significant undertaking but viewing it through the lens of FAIR and FHIR shifts the perspective from “regulatory burden” to “digital transformation”. Organizations that successfully FAIR-ify their data today will not only meet the June 2026 deadline but will also possess a more agile, interconnected, and efficient regulatory infrastructure.
The move to FHIR-structured data is the “hard gate” of 2026. Don’t wait until June to find out your data isn’t Interoperable. Start your FAIR-ification audit today to ensure your submissions are both compliant and reusable. To learn more contact us at info@celegence.com.
Other Related Articles

26 Mar, 2026

10 Mar, 2026

24 Feb, 2026

06 Feb, 2026